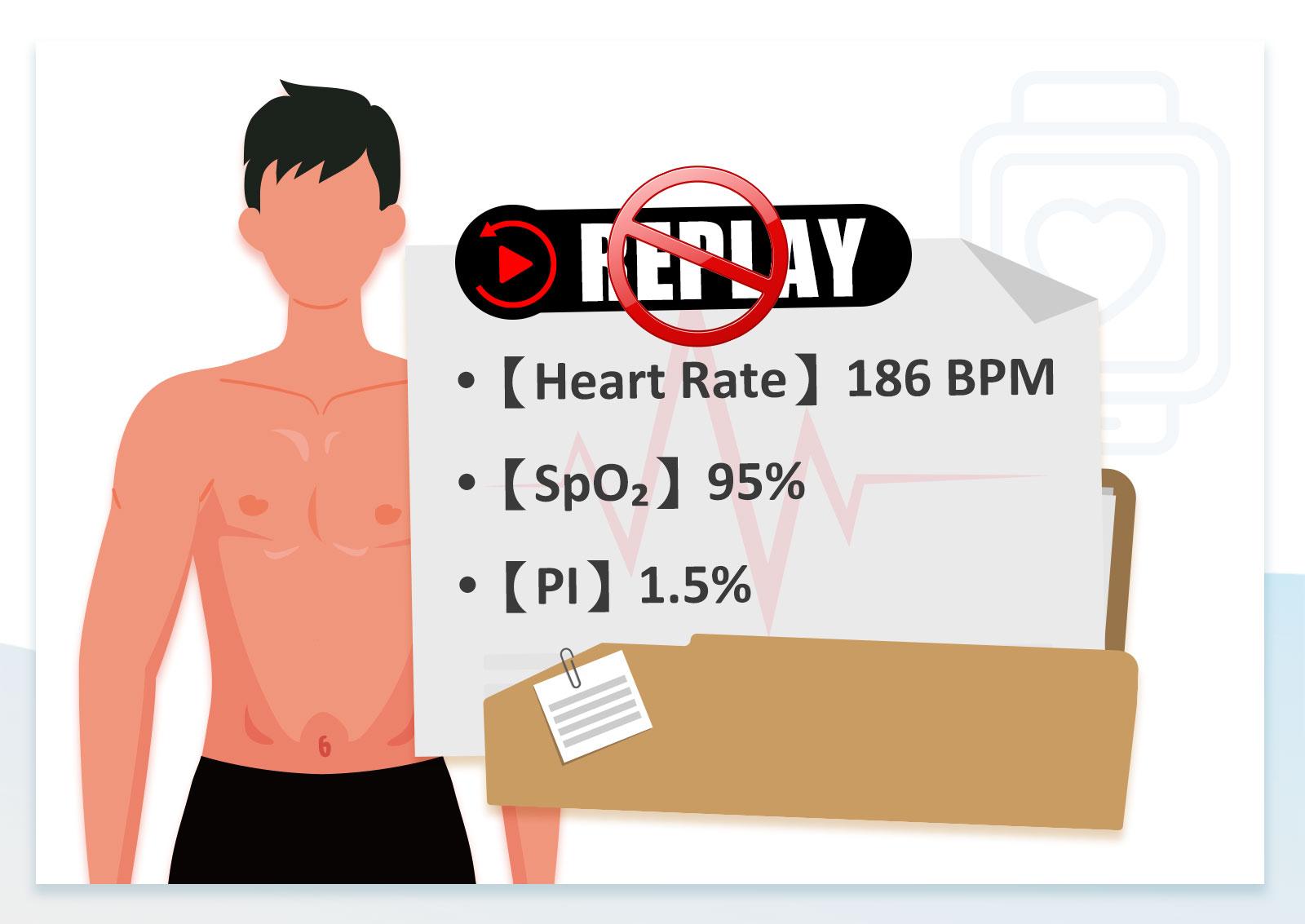
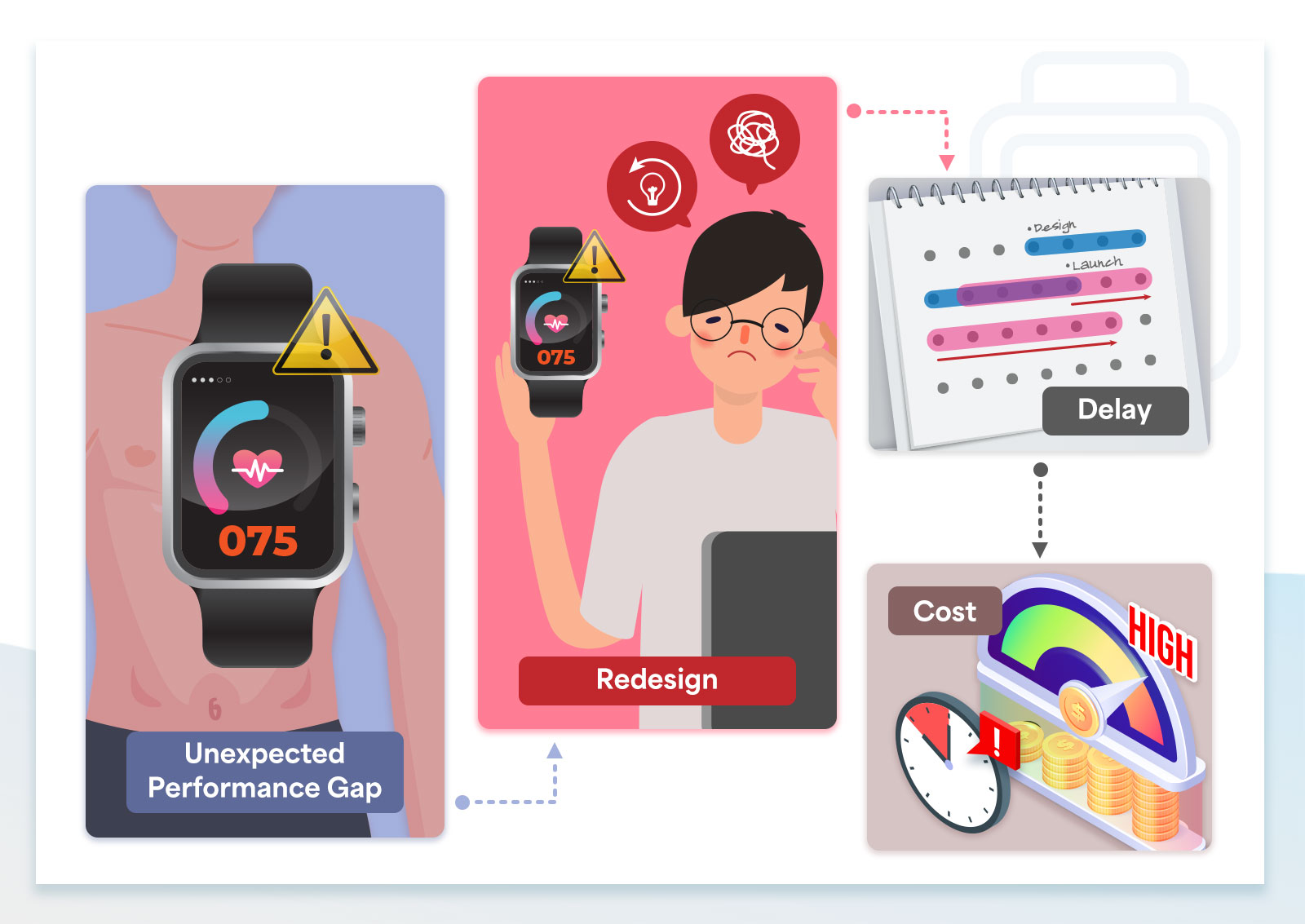
Current testing methods can leave critical performance gaps undiscovered until clinical trials
Performance Testing Under Challenging Physiological Conditions
Conventional test methods struggle to reveal how devices perform in low saturation, low PI, and other hard-to-reproduce conditions.
Inability to Reproduce Identical Test Conditions
The later performance issues are discovered, the harder and more expensive they are to correct.
How PPG simulation fills the Pre-Clinical Test Gap
Validate Full Performance Range Before Human Studies
Validate device behavior across claimed performance ranges under controlled, repeatable lab conditions before human evaluations.
Enable Repeatable Testing Without Relying on Human Subjects
Trusted by Leading Medical Device Teams Worldwide
“Before implementing AECG100, it was difficult to systematically validate our SpO₂ performance range under controlled conditions.
Simulation allowed us to verify edge cases and calibration behavior before entering human evaluation, significantly reducing downstream risk.”
— Eric Hsiao Ph.D., Wearable Biosensing Researcher, Former Biosensor Engineer at Analog Devices




Learn More About the Solution
Solution Overview
PPG Testing Solution at a Glance
Download the 1-page solution overview for key takeaways
Real-World Insight
Learn from an Independent Expert
How a Former ADI Biosensor Engineer Evaluates Reflective PPG Validation Methods
Reference Testing Workflow from Renesas Electronics Corporation
See a Renesas-published example of reflective SpO₂ test setup and procedure using PPG simulation
Technical Fundamentals
From Simulation to Clinical Trials
Building a risk-reduced V&V workflow for wearable PPG devices
How to Test SpO₂ at 70%
Validating low-saturation performance under controlled conditions
Practical Testing Guidance
Application Note — AECG100 (ECG, PPG and PWTT Multifunction Tester)
How to Test Wearable Devices with Reflectance-Based Pulse Oximeter Function