Ensures the quality and accuracy of your products
Medical device manufacturers and medical devices must meet the standards of ISO 13485 and IEC 60601*. As test equipment for medical devices, WhaleTeq products provide customers with the quality and reliability that meet the regulatory requirements.
Measured values may shift after the test equipment is used for a while, so the test equipment needs calibration to maintain its accuracy to carry out precise measurements of products and ensure their quality and stability. Calibration also benefits the test equipment by prolonging its service life with continuous and smooth operations while saving the cost of repeatedly purchasing test equipment from the manufacturer.
At the same time, manufacturers can reduce false passes or false failures in production and maximize productivity by testing products with calibrated equipment. Furthermore, the calibrated equipment may indirectly help avoid the risk of recalling huge amounts of unqualified products resulting in a double workforce, time, money, and even legal disputes.
To comply with medical standards, ensure the quality of medical devices, and reduce the risk of a recall or discards, regular calibration of test equipment is the most convenient and cost-effective option.
*Note: ISO 13485 is an international standard. However, medical device manufacturers are also required to follow the quality control standards of their respective countries, such as QMS of Taiwan, QSR of the U.S., etc.
Manufacturer in-house calibration matters
The manufacturer knows its products best. Like car maintenance and cellphone repair, having experts from the manufacturer conduct calibration is the safest and most quality-assured method.
| In-house Calibration | Third-party Calibration (Outsourced Calibration) | |
|---|---|---|
| Adjustments | O | X |
| A comprehensive inspection of equipment performance | O | X |
| Determination of test results as pass/fail according to the manufacturer's specifications | O | X |
| Complete tests of critical parameters | O | X |
- Not only check the test results but also adjust the test ability to its optimal condition. Through calibrating the entire measurement range, the stable test ability of the equipment is ensured.
- Check the equipment performance in an all-around way to assess whole functions and adjust correspondingly, not just calibrate the equipment after conducting general tests.
- Determine the pass/fail of test results according to the manufacturer's specifications, so the equipment's state is self-explanatory.
- With the manufacturer's specifications, the calibration can focus on test points that tend to shift and be conducted on multiple points to have detailed coverage.
WhaleTeq In-house Calibration benefits you

Reliability
Calibrate to the manufacturer's specifications to ensure performance

Comprehensiveness
Thoroughly inspect the equipment to maintain its performance in the best condition

Traceability
The instruments used for calibration conform with ISO 17025, and TAF/ANAB reports are available to assure the quality and traceable records of WhaleTeq equipment

Efficiency
Complete the calibration in 5 working days to shorten the unavailability for testing
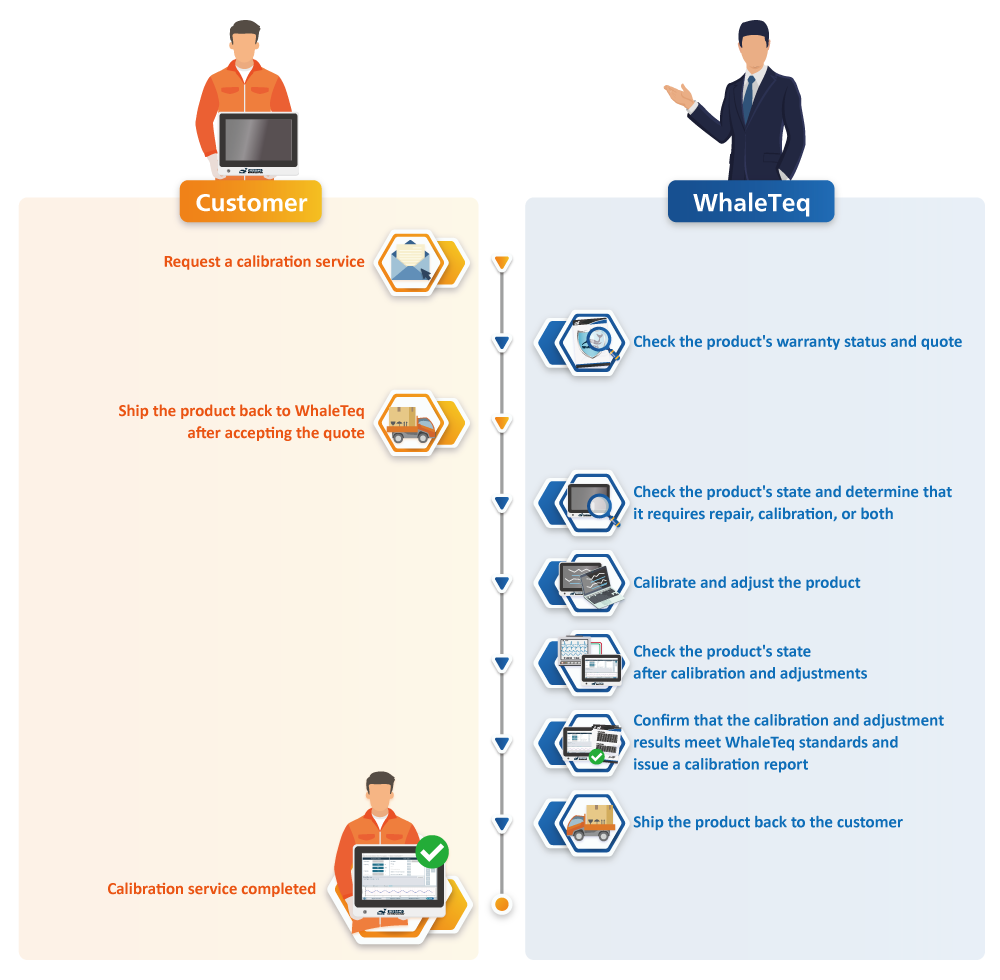
WhaleTeq In-house Calibration Services
Step 1. Check the time range
| Annual | C2 | C3 |
|---|---|---|
| 1-year calibration service | 2-year calibration service | 3-year calibration service |
| Calibrate 1 time within a year | Calibrate 1 time per year | Calibrate 1 time per year |
Step 2. Check whether the TAF/ANAB report is required
| No | Yes | |
|---|---|---|
| WhaleTeq In-house Calibration | Accredited Calibration | |
| Provide TAF/ANAB reports conforming with ISO 17025 (1) | O | |
| The instruments used for calibration conform with ISO 17025 | O | O |
| Calibrate to manufacturer's specifications | O | O |
| A comprehensive inspection of the equipment performance | O | O |
| 5-day calibration service (2) | O |
(1) The working days for calibration are as follows:
● TAF report: about 20 working days
● ANAB report: about 10 working days
(2) The 5-day calibration service starts after WhaleTeq receives the equipment.
Latest News: WhaleTeq Brings Calibration Services to Europe with MPC Romania Partnership!
Supported models:
- ECG series: SECG 5.0 AIO, SECG 4.0, CMRR 3.0+, MECG 2.0, AECG100, WECG400, CMRR 2.0
- EEG series: SEEG 100, SEEG 100E, CMRR 3.0E
Contact:
- MPC Romania: +40 771 293 736
- Email: Daniel Popovici([email protected])
FAQ
Bring Local Product Calibration Services Right to Your Doorstep in the USA!
We're thrilled to announce our recent collaboration with Micro Precision Calibration (MPC) in the United States, bringing local product calibration services right to your doorstep!
Now, our product lines can undergo professional calibration services at MPC, including:
🔹ECG Product Series: AECG100, SECG 5.0 AIO, SECG 4.0, CMRR 3.0+, MECG 2.0
🔹EEG Product Series: SEEG 100, SEEG 100E, CMRR 3.0E
🔹Health Wearable Product Series: WECG400
🔹AED Product Series: DFS100, DFS200, DFS360, DFS400
Contact Person: Alex Leeping <[email protected]>
